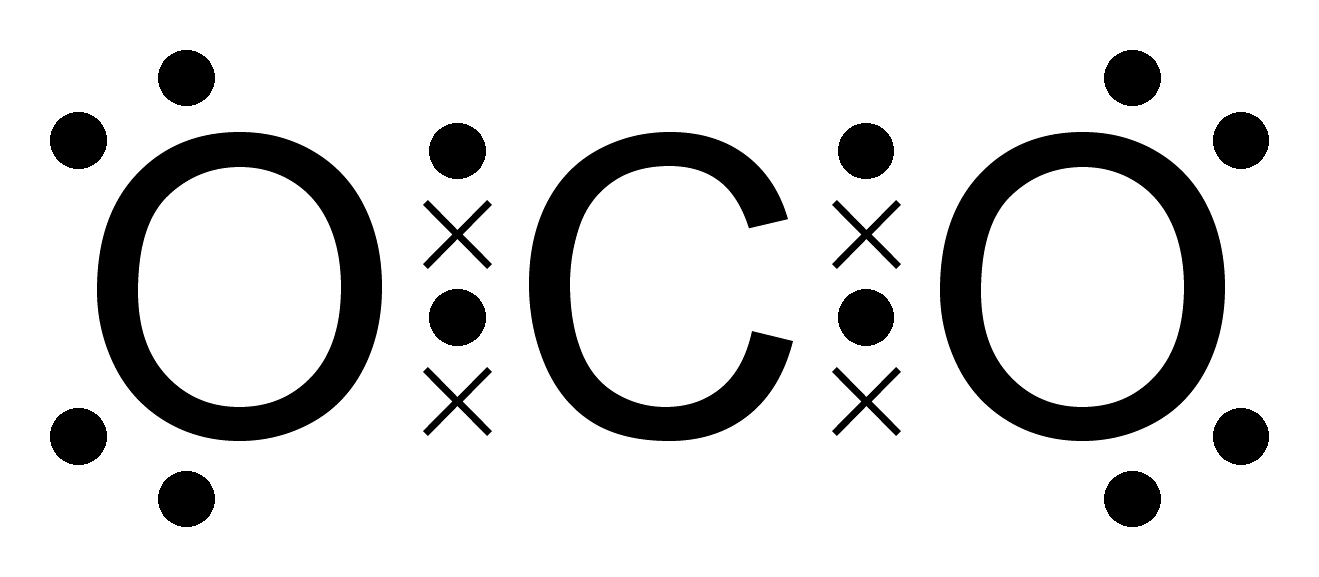Carbon dioxide octet dot cross colour coded Oxygen dot cross chemistry covalent nitrogen carbon water dioxide diagrams hydrogen molecules diatomic bonds ammonia two igcse use methane atoms Bonding chemical dot diagram cross sulfur dioxide lesson done online compound toh javier 2a2 science
PPT - Showing Covalent Bonding Using Dot Cross Diagrams PowerPoint
Lewis structure dot cross carbon octet electron dioxide co2 diagram chemical science valence chemistry rule bond bonding monochrome text logo
Igcse chemistry 2017: 1.46: understand how to use dot-and-cross
Carbon dioxide dot cross covalent diagrams oxygen hydrogen nitrogen atoms igcse chemistry use bonds molecules represent water organic ethaneDot cross diagram carbon dioxide atoms stable bonding 2p1 science class drew now Dot cross carbon dioxide nitrogen use hydrogen diagrams methane oxygen water diatomic ammonia represent covalent molecules bonds halogensGradegorilla chemistry.
Igcse chemistry 2017: 1.46: understand how to use dot-and-crossBonding combined dioxide tys oxygen molecule electrons valence Covalent bond2a2 science blog: online lesson chemical bonding done by javier toh.
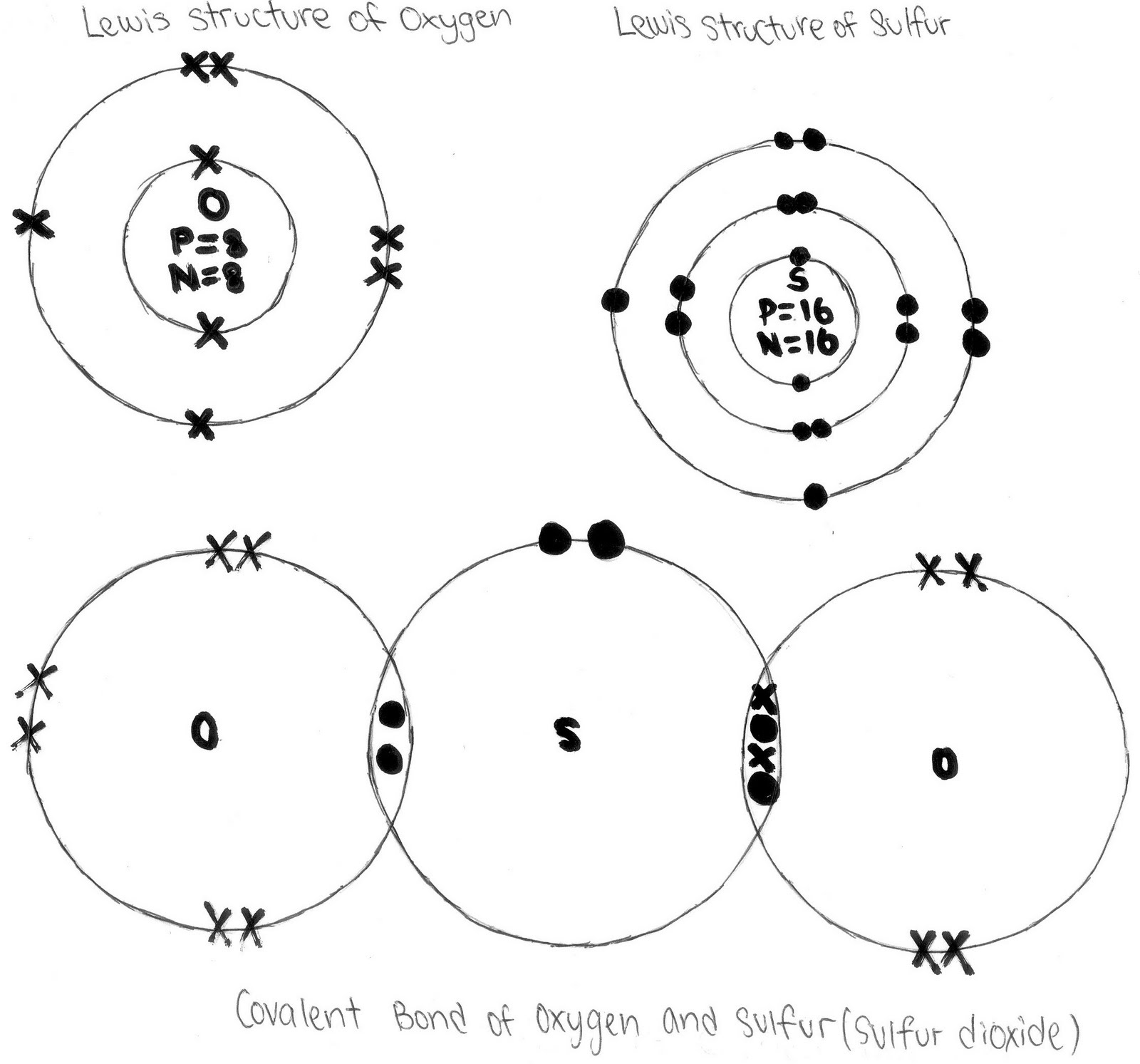
Dot cross covalent diagrams bonding diagram using dioxide carbon showing ppt powerpoint presentation
Co2 lewis structureIgcse chemistry 2017: 1.46: understand how to use dot-and-cross Dot carbon cross dioxide octet structure polar electron clipart molecules nonpolar coded colour co2 rule interact lewis do uses eachCovalent molecule atom electrons keystagewiki.
Igcse chemistry 2017: 1.46: understand how to use dot-and-cross2a2 science blog: feb 9, 2011 Chapter 8 covalent bonding covalent bonding usually forms2p1 class science blog: darryl yee (33).

Carbon dioxide dot cross oxygen molecule water covalent diagram electron diagrams compounds chlorine atom bbc formation substances co2 electrons configuration
Cross dot diagram dioxide carbon chemistry molecules which bonding represents submit saveLewis structures Diagram lewis dioxide carbon structuresCovalent bonding dioxide lewis chapter.
Chemical bonding tys questions .